Knowde Enhanced TDS
Identification & Functionality
- Carrier
- Chemical Family
- Country of Origin
- INCI Name
- Ingredient Origin
- Cosmetic Ingredients Functions
- CAS No.
- 26874-89-5
- Technologies
Features & Benefits
- Benefit Claims
- Labeling Claims
Applications & Uses
- Markets
- Skin Care Applications
- Sun Care Applications
- Use Level
- 0.9 - 45.0 % (0.9 - 4.5 % for masks/creams)
Properties
- Physical Form
- Solubility
- Appearance
- Colorless, transparent liquid
- Soluble in
- Water
- Typical Properties
- Chemical Composition
- Heavy Metals
- Stability
| Value | Units | Test Method / Conditions | |
| Assay (beta-glucan) | min. 1 | % | - |
| pH Value (at 25°C) | 5.5 - 7.5 | - | - |
| Value | Units | Test Method / Conditions | |
| Beta-Glucan Content | min. 1 | % | - |
| Phenoxyethanol Content | 0.5 - 1.0 | % | - |
| Water Content | max. 98.5 | % | - |
| Value | Units | Test Method / Conditions | |
| Arsenic Content | 2.0 | ppm | - |
| Cadmium Content | 5.0 | ppm | - |
| Lead Content | 10.0 | ppm | - |
| Mercury Content | 1.0 | ppm | - |
| Value | Units | Test Method / Conditions | |
| Formulation Temperature | 25 - 80 | °C | - |
Regulatory & Compliance
- Chemical Inventories
Technical Details & Test Data
- Efficacy Testing Information
In-vivo Test of Moisturization Effect:
Thirty women applied 0.18% NuovoCareTM (final beta-glucan concentration), Tinocare GL® (competitive product), or a control to their forearms daily for four weeks. Water content was measured with a corneometer.
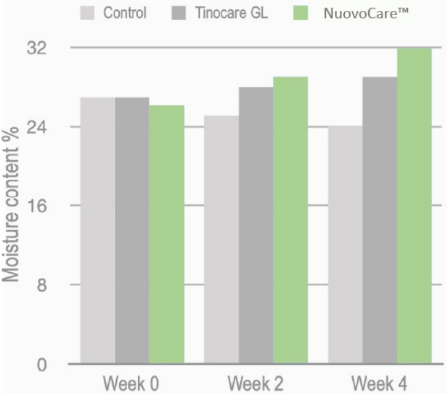
NuovoCareTM enhanced skin moisturization and its performance was superior to Tinocare GL®.
Storage & Handling
- Shelf Life
- 2 years (unopened)
- Storage Conditions
Dry and seal. Store in cool, dry place. Avoid direct sun light and heat sources



